Projection of plot to planes in 3D graph.Skip points with option to maintain overall data features.Connect lines across axis break or missing data, etc.Show or hide plot, all plots of same name, all plots in a layer.Change X or Y of the plot, drag to add new plot, remove plot, and change plot orders. 
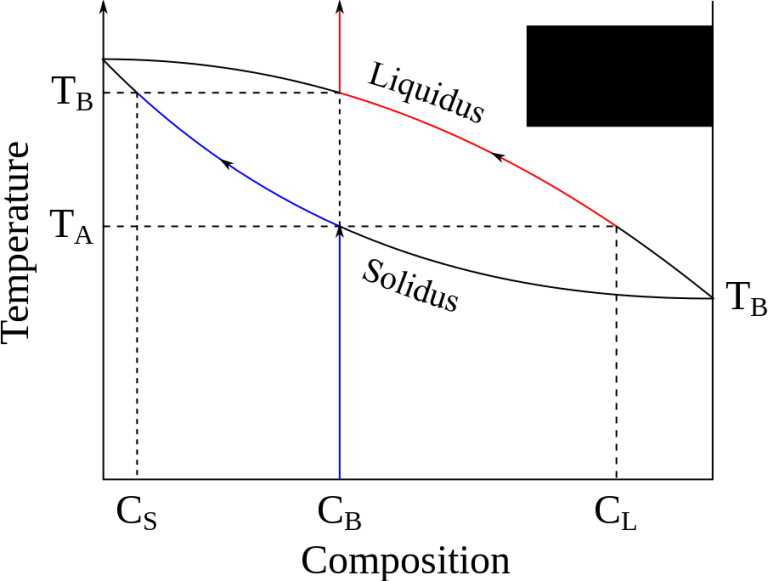
Change exist plot type and mix different plot types in one graph.Based on plot type, customize plot attributes such as symbol shape and size, line style and width, column and area pattern, transparency, etc.OriginPro page to view a comparison table. Features specific to OriginPro are marked with the PRO icon in this page. In addition to all of Origin's features, OriginPro offers advanced analysis tools and Apps for Peak Fitting, Surface Fitting, Statistics and Signal Processing. Take your data analysis to the next level with OriginPro. Connect with other applications such as MATLAB™, LabVIEW™ or Microsoft© Excel, or create custom routines within Origin using our scripting and C languages, embedded Python, or the R console. Extend the capabilities in Origin by installing free Apps available from our website. Origin graphs and analysis results can automatically update on data or parameter change, allowing you to create templates for repetitive tasks or to perform batch operations from the user interface, without the need for programming. Origin offers an easy-to-use interface for beginners, combined with the ability to perform advanced customization as you become more familiar with the application. Origin is the data analysis and graphing software of choice for over half a million scientists and engineers in commercial industries, academia, and government laboratories worldwide. Browser Graph for Multichannel Data Exploration.Float Windows outside of Origin Interface.Extended Templates from OriginLab Website.w l is the mass fraction of the whole sample in the liquid phase. The same equations can be used to find the mass fraction of alloy in each of the phases, i.e. The tie line drawn is from the solid alpha to the liquid and by dropping a vertical line down at these points the mass fraction of each phase is directly read off the graph, that is the mass fraction in the x axis element. There is now more than one two-phase region. Then the liquid concentration will start increasing.Įutectic phase diagrams Tie line in the Alpha plus Liquid two phase region If you're having difficulty realising why this is so, try visualising the composition when w o approaches w l. And then the denominator is the overall length of the arm so the difference between the solid and liquid compositions. That is if you want the mass fraction of solid then take the difference between the liquid composition and the original composition. The numerator of each equation is the original composition that we are interested in is +/- the opposite lever arm. Where w B is the mass fraction of element B for the given composition (represented as w o in this diagram). In an alloy or a mixture with two phases, α and β, which themselves contain two elements, A and B, the lever rule states that the mass fraction of the α phase is It can be used to determine the fraction of liquid and solid phases for a given binary composition and temperature that is between the liquidus and solidus line. In chemistry, the lever rule is a formula used to determine the mole fraction ( x i) or the mass fraction ( w i) of each phase of a binary equilibrium phase diagram. Formula for determining the mole or mass fraction of phases in a binary phase diagram
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
